The dimensions of the capillary show that this kind of apparatus is designed for medium- to low-viscosity substances: mineral oils, paper coatings, and dispersions, and for polymer solutions used to produce synthetic fibers on spinning machines. Shear stress values, however, do not exceed the medium range (a typical value would be 25 kPa). This method can operate in a shear rate range up to 1,000,000 s -1. Gas forces the sample through the capillary at a preset pressure. The inner diameter lies between 0.2 mm and 1 mm, while the length ranges from 30 mm to 90 mm. These devices feature a glass or steel capillary of exact inner diameter and length. Capillary viscometers utilizing gas pressure Typical applications are highly viscous substances, such as polymer melts, and also PVC plastisols, greases, sealants, adhesives, and ceramic masses. However, the weight is replaced by a driving motor, which realizes high shear stress values (up to 900 kPa) and medium to high shear rates (around 1,500 s -1). Such a viscometer works in the same way as a weight-driven instrument (see the above section). High-pressure capillary viscometers with electric drive In such devices, the polymer sample is exposed to medium shear stresses (from 3 kPa to 200 kPa) and medium shear rates (from 2 s -1 to 200 s -1).
#HIGH VS LOW VISCOSITY ISO#
ISO 1133 states the dimensions of cylinder, piston, and die as well as the approved weight pieces. capillary) at the bottom of the cylinder. The sample then has to pass through an extrusion die (i.e. The steel piston glides down inside a vertical steel cylinder, which contains the sample. Ī defined weight on top of a piston is pulled down by gravity. Consequently, such an instrument is also named MFR or MVR tester. These parameters help to assess the quality of the melt and to predict its behavior when processing it.

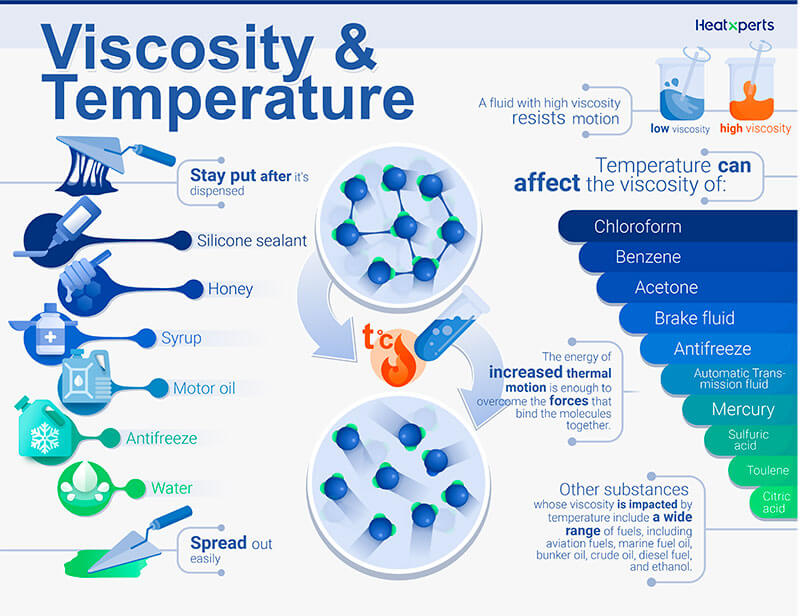
The device returns the MFR (melt mass flow rate) in or the MVR (melt volume flow rate) in. The video below shows several different long chained oils, each progressively more viscous.This type of instrument plays a major role in checking polymer melts. Glycerol, CH 2OHCHOHCH 2OH, is viscous partly because of the length of the chain but also because of the extensive possibilities for hydrogen bonding between the molecules. Fuel oil, lubricating grease, and other long-chain alkane molecules are quite viscous for this reason. This is because the molecular chains get tangled up in each other like spaghetti-in order for the liquid to flow, the molecules must first unravel. Liquids containing long molecules are invariably very viscous. Honey, mostly glucose and fructose (see image below) is a good example of a liquid which owes its viscosity to hydrogen bonding. Liquids whose molecules are polar or can form hydrogen bonds are usually more viscous than similar nonpolar substances. Viscosity is governed by the strength of intermolecular forces and especially by the shapes of the molecules of a liquid. Those like ether or gasoline which flow very readily have low viscosities. Liquids which flow very slowly, like glycerin or honey, have high viscosities. The resistance to such flow is called the viscosity.
Because its molecules can slide around each other, a liquid has the ability to flow.
